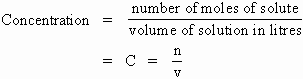
Concentration
Previous Page: Reacting quantities...
Concentration (not how hard you are thinking) has its opposites in dilute and concentrated. This was discussed previously in the Chemistry of Solutions unit, which you may find useful to revise at this point. Take a set volume of solution, say 200 millilitres of hot water. Add to it a small amount of, for example, tea. It colours slightly. A small amount of tea has dissolved in the water. The tea is dilute. Now pop the tea bag back in for ten minutes. Now the tea solution is highly coloured. The tea is concentrated.
Many of the substances we deal with in the laboratory (or indeed in everyday life) are not in solid form, rather they are dissolved in water. So it would be handy if we were able to deal directly with solutions by measuring the volume of solution present and how much solid (solute) was dissolved in the solvent (water).
Concentration, c, is commonly expressed as amount of solute (substance dissolved) over volume of solution (not solvent). If amount of solute is measured in moles, n, and volume, V, is in litres, then concentration, in moles per litre (mol L-1) is called molarity.
We can develop a formula that will enable us to calculate concentration of a solution simply by using our definition.
The formula is:
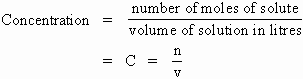
1 mole of NaCl has a mass of 58.44g. If 58.44g of NaCl is dissolved in enough water to make up one litre after the NaCl is added and dissolved, the concentration of the solution is said to be one molar and is written as 1M or 1mol L-1. If 29.22g of NaCl is dissolved in enough water to make up one litre, the concentration of the solution is 0.5M or 0.5mol L-1. If 58.44g of NaCl is dissolved in enough water to make up two litres, the concentration of the solution is 0.5M or 0.5mol L-1 and so forth.
![]()
Example 1: What is the concentration of a solution with 100g CaCO3 in 1 litre?
Solution: One mole of CaCO3 contains:
1 mole of Ca
1 mole of C
3 mole of O.
| 1 mole of calcium is | 40g | ||
| 1 mole of carbon is | 12g | ||
| 1 mole of oxygen is | 16g, so | 3 mole is | 48g |
| 1 mole of CaCO3 is | 100g |
![]()
(Isn’t that tricky?)
![]()
Example 2: What is the concentration of a solution with 100g CaCO3 in 500mL?
Solution:
![]()
Example 3: How much KMnO4 would I need to make 100mL of 0.01M solution?
Solution: One mole of KMnO4 contains:
1 mole of K
1 mole of Mn
4 mole of O.
| 1 mole of potassium is | 39.1g | ||
| 1 mole of manganese is | 54.9g | ||
| 1 mole of oxygen is | 16g, so | 4 mole is | 64.0g |
| 1 mole of KMnO4 is | 142.0g |
We want mass, so first must find moles, n.
C = n/V becomes n = CV = 0.01mol L-1 × 0.1L = 0.001mol.
![]()
Example 4: What volume of KMnO4 could I make if I needed a 0.1M solution and had only 7.1g of solid KMnO4?
Solution:
![]()
![]()
Now try these problems.