You MUST be clear on the meanings of the terms concentrated, dilute, strong and weak.
Concentrated and dilute are not particularly technical terms. They have pretty much the same meaning in acid-base chemistry as in everyday usage. Dilute refers to a solution with lots of solvent and not so much solute (lots of water, little salt). Concentrated is the reverse.
Strong and weak are entirely distinct. They refer to degrees of dissociation. A substance that dissociates, or breaks up in solution, to a large degree is strong. A substance that dissociates to a small degree is weak. When mixed with water, HCl ionises completely (that is, about 100 out of 100 molecules donate their hydrogen ion). Another way to say exactly the same thing is HCl dissociates completely to H+ and Cl- ions. (Even that is somewhat redundant, but never mind.) Ethanoic [acetic] acid, on the other hand, dissociates only slightly (that is, about 1 out of 100 molecules separates from its hydrogen ion). In an acetic acid solution, there is a large number of CH3COOH molecules, and just a few H+ and CH3COO- ions. HCl is a strong acid, CH3COOH is a weak acid.
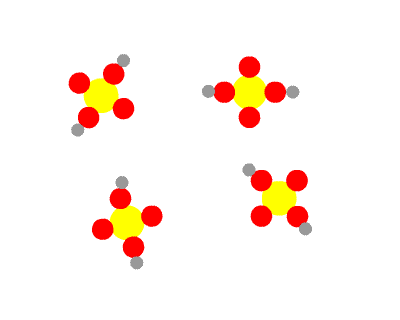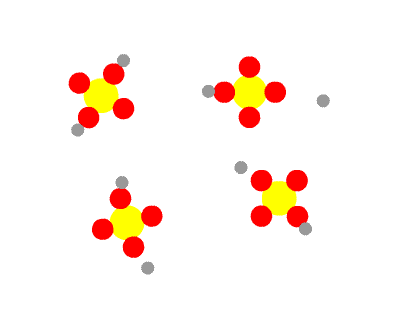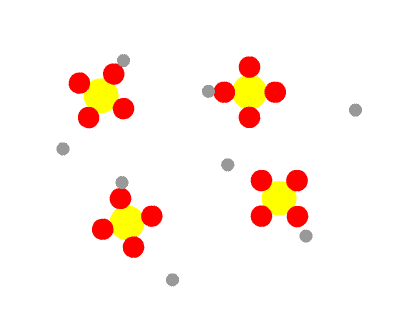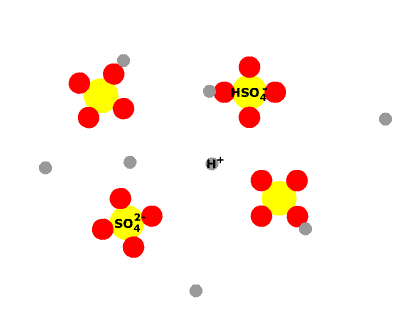
Sulphuric acid, H2SO4 is a diprotic acid, having two protons to donate. The first is strong, the second weak. This means that in a solution of H2SO4 you would find no H2SO4 molecules, all having dissociated into H+ ions and HSO4- ions. Some of the HSO4- ions further dissociate into H+ ions and SO42- ions, so three species would be present in the end.

As a consequence, HSO4- is amphiprotic. It can donate a proton, the behaviour of an acid, as it splits into H+ ions and SO42- ions. Under certain circumstances, it can accept a proton, the behaviour of a base, reconverting to H2SO4.
It should now be evident that it is completely normal to have a concentrated, weak acid or a dilute, strong one. The same is true for bases and any other species that dissociate (including most ionic substances, eg NaCl). In fact, there are distinct correlations between strong and soluble, and between weak and insoluble.
This brings us to a discussion of describing degrees of acidity. Acidity is measured in units called pH. The literal translation of pH is negative logarithm of the hydrogen ion concentration. Recall that the log10 of 10 is 1, the log10 of 100 is 2, the log10 of 1000 is 3, and so on. Also, the log10 of 0.1 is -1, the log10 of 0.01 is -2, the log10 of 0.001 is -3,and so on. Thus, the negative log10 of 0.001 is 3, the negative log10 of 0.01 is 2, the negative log10 of 0.1 is 1. That is; the higher the number, the lower its negative log. Higher concentration = lower pH. This means that a solution measuring pH = 1 is more acidic (contains more hydrogen ions) than a solution measuring pH = 5.
The term concentration normally refers to dilute or concentrated, but strong and weak also can affect the pH. Think again about HCl and CH3COOH. A dilute solution of HCl will have a lower concentration, so a higher pH number, than a concentrated one. The same is true for CH3COOH. But with equally concentrated solutions of HCl and CH3COOH, the concentration of hydrogen ions in the HCl will be greater because it is a stronger acid, so its pH number will be lower. Thus both concentration and acid strength affect the acidity of a solution.
Next Page: Typical Reactions