Aqueous Solutions and Precipitates Previous Page: Solutions
In particular we are concerned with water-based solutions.
 | One of the goals of alchemists, aside from trying to transmute lead into gold, was to find the universal solvent (and the container to store it in. Think about it). In fact, if you are patient enough, the universal solvent is well known - it's H2O. Have a look at Cataract Gorge. All the rock that has been removed to form the gorge was dissolved by water, and the process continues today. That's what I mean by being patient. |
In a more practical sense, most atoms and compounds are classified as soluble or insoluble in water. This is done for simplicity, because in fact, some compounds are very soluble, some are pretty soluble, some are fairly soluble, some are slightly soluble and some are marginally soluble, like the dolerite in the gorge. You can see that trying to classify substances simply as either soluble or insoluble is a brave undertaking.
Over time people have built up a knowledge base recording which ionic compounds are likely to be soluble, and under what circumstances; and organised this information in a table, the Solubility Table. All sodium compounds are soluble. All potassium compounds are soluble. All ammonium compounds are soluble. All nitrates are soluble. (All acetates [ethanoates] are soluble - but they rarely come up.) HINT: It is often easier to memorise Na+, K+ and NO3- as always soluble. Saves you having to look at the Solubility Table all the time. (The Solubility Table lists rules to predict whether an ionic compound will be soluble or not in a particular situation.)
| Question: Do we expect sodium chloride to be soluble in water at moderate temperatures? Answer: Find Cl- in the table. Chlorides are generally soluble so we generally expect ionic compounds containing chloride ions to be soluble in water at moderate temperatures. Sodium is no exception so we take sodium chloride to be soluble under the conditions described. | .gif) |
| | | | | NaCl(aq) | AgNO3(aq) | NaCl(s)+ AgNO3(aq) | | How about silver nitrate? NO3- is found in the table, which suggests that ALL nitrate compounds are soluble. Therefore we can assume that sodium chloride is soluble. We can assume that silver nitrate is soluble. What happens if we mix solutions of the two? |
First, recognise all these substances from the Table of Common Ions. We are dealing with ionic compounds as we discuss solubility. This is not by definition. Covalent and metallic substances may also be involved in solutions. Ionic compounds demonstrate these concepts most clearly.
Having said that, the number of things that might happen are actually few.
- The first possibility is nothing. No reaction occurs, no evidence of chemical change is observed.
- The second possibility is a chemical reaction. You may observe a colour change, a precipitate forming, water condensing on the glass, bubbles, hissing, vile odours, flashes of light, peals of thunder, the room spinning before your eyes, etc, etc.
In the case of sodium chloride and silver nitrate a precipitate forms. What could it be? Since it is an ionic substance it cannot be a compound of silver and sodium (two positive ions), nor of nitrate and chloride (two anions). Since it wasn't present in the original separate solutions it cannot be a compound of sodium chloride or silver nitrate. This leaves only two possibilities: it may be sodium nitrate or it may be silver chloride. Given the initial solutions THESE ARE THE ONLY TWO POSSIBILITIES.
| Since we have already assumed from the table that nitrates are soluble without exception, the evidence seems to be pointing strongly to silver chloride. Checking again the table we discover that chloride compounds are generally soluble, but silver chloride is listed as an exception. Thus silver chloride is confirmed as the most likely identity of the white solid (the precipitate) that is by now sitting on the bottom of the test tube. (Other possibilities involve either contaminants or violation of the solubility agreement.) | 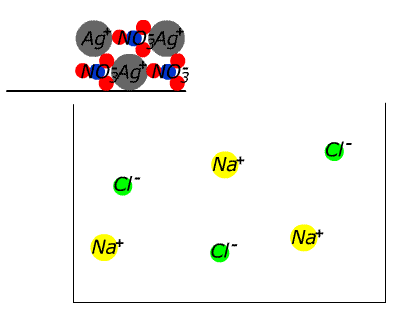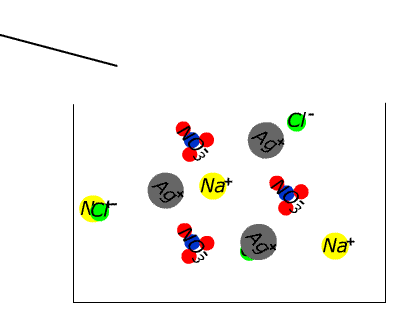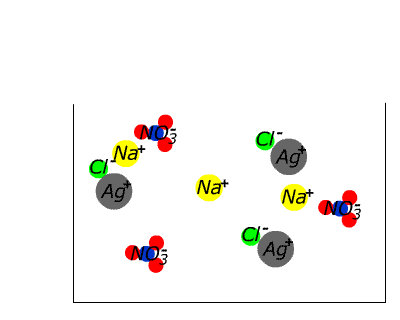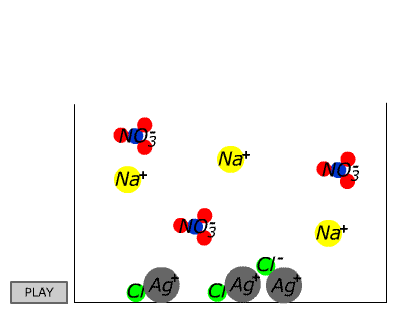 |
Next Page: Acids and Bases
Link to Index

.gif)



