Electrons: pooled, donated or shared?
In an earlier segment you learned that atoms of metallic elements were not too fussed about pooling their least-attracted electrons. You will recall that opposite charges attract, so positive protons attract negative electrons. This electrostatic attraction is one of the four fundamental forces of nature, which are studied in far greater detail in physics. Suffice to say, the electrostatic attraction, or electromagnetic force, to be more correct, is stronger than gravity but weaker than the nuclear forces that keep the positive protons in the nucleus from flying apart, and hold the uncharged neutrons there as well.
The electrons, as they whirl around outside the nucleus, are attracted to the protons inside. The closer the electron, the stronger the attraction. (A similar situation holds with gravity. You are more strongly attracted to the earth than is an astronaut aboard a space station.)
Thus it is that the electrons in the outermost shell find it easiest to leave, and become part of the electron pool in a metallic bond.
These electrons are often called valence electrons. They are the electrons in any given atom that are responsible for its bonding to other atoms.
In another segment we extended this model to ionic substances. A metallic atom can, in fact, donate (give up influence over) one or more loosely held electrons to a non-metallic atom. The metallic atom gains charge, the non-metallic atom gains an electron. The former becomes a positive ion, the latter a negative ion. Opposite charges attract and you get an ionic compound.
This discussion may lead you to wonder how, or even whether, bonds form between non-metallic atoms.
Notice that the word molecule has not yet been introduced. If you were taught previously that molecules were groups of joined atoms you were not given the whole picture (even as you are not being given the whole picture now, only more of it).
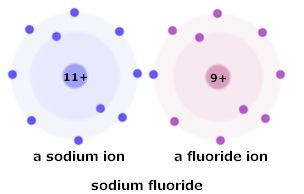
Recall once more the sodium and fluorine atoms discussed earlier. Sodium is happy to shed its outermost, least attracted electron. Fluorine would love to gain one. Both would then have the full outer shell they aspire to. (This is, of course, anthropomorphism gone berserk.) They can then be electrostatically attracted ions and form an ionic compound, NaF. It's a nice simple picture, and in the case of NaF the reality is nearly that simple, except that in any real situation it would happen on a scale of very, very big numbers.

But consider the compound F2. Single atoms of fluorine are very rare because a fluorine atom wants very badly to be joined to another atom - any other atom. In part, this is because fluorine has an opening in its outer energy shell, and in part, because that opening is close to the nucleus. This arrangement allows lots of positively charged proton attraction to reach out, and not have to reach very far. Say two reactive fluorine atoms just happen to bump into each other. Each tries to grab a valence electron from the other, as was seen with sodium. But this time neither is at all inclined to give up an electron. So they stick together, form a bond, become F2. This is, admittedly, a simplistic description, but I think it puts across something of the idea about what is actually going on. We usually say those electrons grabbed each by the other fluorine atom are shared. The corresponding shell diagram looks like this.
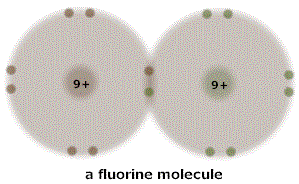
Note that bonds formed by shared electrons ALWAYS involve pairs of electrons. Ionic bonds may, but are not required to.
Next Page: Kinds of Covalent Compounds
Related Topic: Predicting Bond Type