Drawing Simple Alkanes
(semi-structural formulae)
Previous: Naming Simple Alkanes
If I were to draw a methane molecule it might look like this:
CH4.
Ethane and propane could be drawn thus:
CH3-CH3 and CH3-CH2-CH3
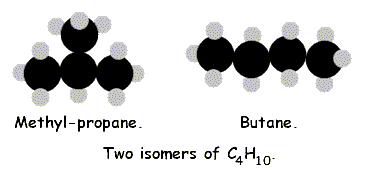
The next molecule in the series has the molecular formula C4H10 , so might look like this:
CH3-CH2-CH2-CH3
or this:
| CH3-CH-CH3 | ||
| | | ||
| CH3 | ||
The first is, indeed, butane. The second is called methyl propane. That is, it is propane because its longest chain of carbon atoms is three carbons in a row (prop-), but it has a side chain that is one carbon long (meth-). The two molecules have the same molecular formula, C4H10, but different structural formulae, as shown. Therefore they are isomers.

The next molecule in the series has the molecular formula C5H12.
|
|
| |||||||||||||||||||||||||||||||||||||||||||||
All three of these compounds fit the molecular formula. They are, left to right, pentane, methyl butane and dimethyl propane. Again, they are all isomers of each other. If you have a 3D model of dimethyl propane you can see that it makes no difference, indeed, no sense, to worry about which carbons are the ends of the three carbon chain, and which carbons are the methyl side chains.
Next:First Naming Rules
Link to Index