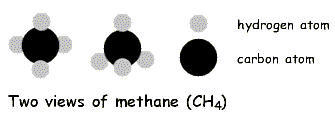
Naming Simple Alkanes
The naming of simple alkanes is a simple matter. The naming of complex alkanes is a complex matter. We will restrict our concerns to the former.
I suspect the main difficulty in this process is the same as in map making - you are really presenting a three dimensional object on a two dimensional surface. It will be quite useful to avail yourself of a standard molecular model kit, if you can find one.
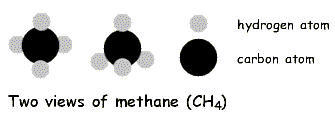
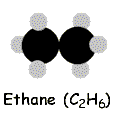
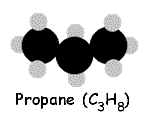
Bearing that in mind, the simplest alkane is methane, which has the molecular formula CH4. Next is ethane, with the formula C2H6. Then propane, C3H8 and butane, C4H10. And so forth.
Alkanes have the generic formula CnH(2n+2), as you can see in the table of the first ten alkanes below.
Number of carbons in chain | Formula for alkane | Formula for branch | name |
1 | CH4 | -CH3 | methyl as in methane |
2 | C2H6 | -CH2-CH3 | ethyl as in ethane |
3 | C3H8 | -CH2-CH2-CH3 | propyl as in propane |
4 | C4H10 | -CH2-CH2-CH2-CH3 | butyl as in butane |
5 | C5H12 | -CH2-CH2-CH2-CH2-CH3 | pentyl as in pentane |
6 | C6H14 | (-CH2-)5-CH3 | hexyl as in hexane |
7 | C7H16 | (-CH2-)6-CH3 | heptyl as in heptane |
8 | C8H18 | (-CH2-)7-CH3 | octyl as in octane |
9 | C9H20 | (-CH2-)8-CH3 | nonyl as in nonane |
10 | C10H22 | (-CH2-)9-CH3 | decyl as in decane |
Next: Drawing Simple Alkanes