Refresh to replay. Close window to resume. Text continues below animation.
strong | weak | |
concentrated | 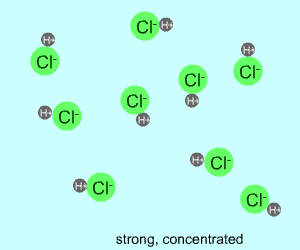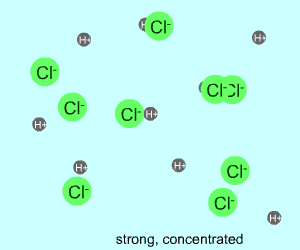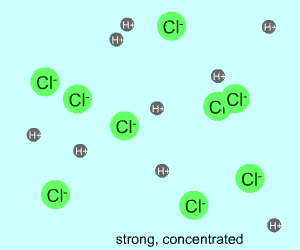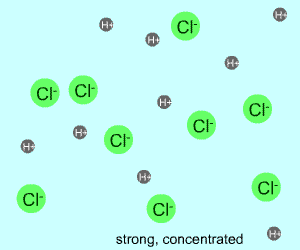 | 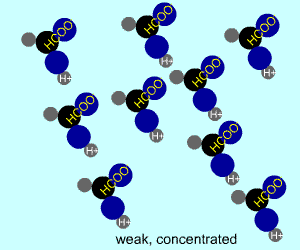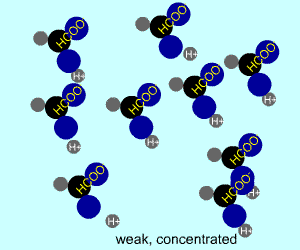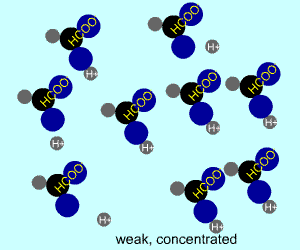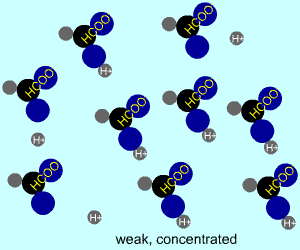 |
dilute | 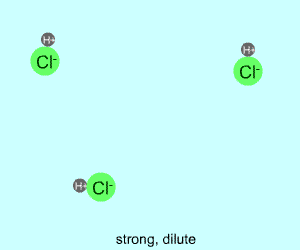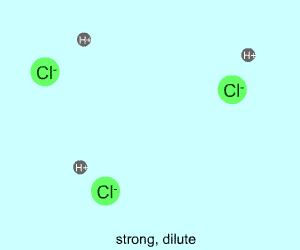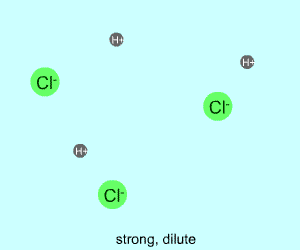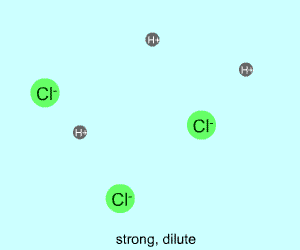 |  |
Strong v Weak v Concentrated v Dilute
Refresh to replay. Close window to resume. Text continues below animation.
strong | weak | |
concentrated |  |  |
dilute |  |  |
Strong v Weak v Concentrated v Dilute
Any electrolyte (acid, base, salt) has a characteristic degree of dissociation (breaking apart). With acids in particular, this is a central concept as the degree of dissociation has a large effect on the pH of a solution. The pH of a solution reflects the concentration of hydrogen ions (H+). One way to get lots of these in a solution is to have lots of acid molecules in the solution. The other way is to have molecules that provide lots of hydrogen ions.
The table above shows the four possibilities: high and low concentration and high and low dissociation. Note that the window that is high on both counts (upper left) has lots of ions (H+), therefore the lowest pH. The window that is low on both counts (lower right) has very few ions, so a higher pH number. The other two windows have roughly the same number of ions, so a middling pH number. In real life either could be greater than the other; this is only an approximation.