



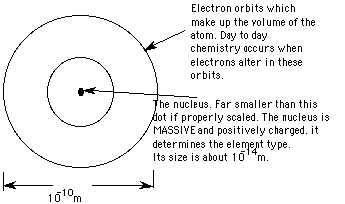
Imagine the Melbourne Cricket Ground. A cricket ball is being hit in the centre. Shrink it all to one millionth of a millimetre.
The MCG is now an atom, the cricketball a nucleus.
The things wrong with this is where all of the mass is. If it were an atom, the cricket ball would have virtually all of the mass, the MCG nearly none at all and the cricket ball is too large, a small marble would have been better.
What evidence is there for such a strange thing? After all, no one has actually SEEN an atom nor are they likely to, yet we can now display "pictures" of what we assume are atoms when rather peculiar measurements with electrons are carried out. Indeed we can move around individual atoms. These " best " pictures show up as fuzzy blobs, not electrons and nuclei. The microscopes to do this are NOT electron microscopes but machines called Atomic Force microscopes and Scanning Tunneling microscopes.
We can even make movies with them!
Our evidence comes from hitting atoms as hard
as we can with "α
radiation or other pellets. When this is done the radiation bounces off
- "scatters" -in a manner only consistent with this model. The MCG picture
(model) is now very well established having been around for about 70 years.
The atom then, can best be pictured as sort of
like a solar system, the "planets" are electrons, minuscule things
with a negative electrical charge, the "sun" is the nucleus, tiny,
massive and positively charged. The whole thing is kept together by electricity
- electrostatic attraction between opposite charges, rather than gravity.
The outside of the atom - the electrons - cause the day to day reactions of chemistry. They jump about in their orbits giving off light or absorbing it , rearranging themselves in chemical reactions to create molecules, or even to leave, causing the atom to become a positive ion!



Images of atoms
The nucleus is unaffected by all of this carry on. It carries the identity of the atom - whether it is sodium or oxygen or uranium. If it is going to change, it will do so in a BIG WAY, typically a million times more energetically than anything the electrons can do.
The nucleus itself has two main components;
PROTONS; Far more massive than electrons (1860 times ), protons have a mass of 1.67 x 10-27 kg. They have a single elementary positive charge meaning that their charge is exactly equal to but opposite to that of the outer electrons.
The number of protons ( the ATOMIC NUMBER, Z ) decides the atom type.
NEUTRONS; Very slightly more massive
than the proton but it has no electrostatic charge at all. With the exception
of hydrogen, most of the elements have an equal or larger number of neutrons
in the nucleus to the number of protons.
Two otherwise IDENTICAL atoms may have different numbers of neutrons. When this occurs we speak of ISOTOPES of the elements.
To identify the isotope that concerns us unambiguously, we write the element giving the Atomic Number and the MASS NUMBER, A.
The mass number is the total number of protons and neutrons to be found in the nucleus.
We refer to protons and neutrons together as
NUCLEONS.
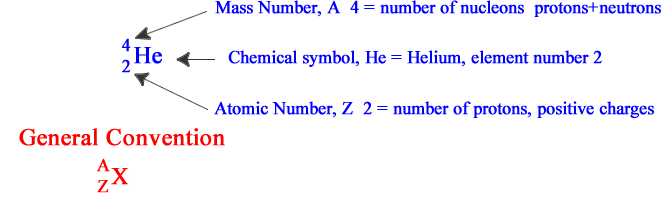
Phosphorus has many isotopes -
![]()
( A further complication is the Atomic Mass used by Chemists. This is an averaged version of the Mass Number over the various proportions of natural isotopes. Chlorine exists naturally as both Cl-35 and Cl-37 in the proportions 75% to 25% so the Atomic Mass is 35.5.)
Isotopes of an element behave chemically identically.
This is because they have the same Atomic Number, so the same electron structure.
The implication is that unstable isotopes will behave, even in biological situations, the same way as the stable form of the atom. This can be useful in medicine, but is dangerous in "fallout".
The slight differences in physical properties that do occur can be exploited for measurements or separating particular isotopes.
Use of O-18 in geology and climatography
Past climatic history is reflected in the biological take up of oxygen-18 as opposed to oxygen-16, the most common isotope of oxygen. This uptake is temperature sensitive. In warmer conditions the uptake ratio is different to colder conditions due to different densities of the gases.
Uranium-235 separation by ultracentrifuges or
other techniques also rely on the slight differences in density that the
different numbers of component neutrons make.
NUCLEAR STABILITY
We laugh at the notion of the alchemists' dream of turning lead into gold. How absurd we tend to think, yet it can be done! Certainly not by chemistry, but by altering the nuclei of atoms we can create artificial atoms.
All of the world's stock of americium, plutonium ( tonnes of it ) and medicinal isotopes including cobalt - 60 are artificial, made in accelerators or reactors. They did not exist before this century on Earth.
Why don't these exist naturally?
The elements of which the Earth is made are thought to have been created in a star existing previously to our own. This star went supernova - a colossal explosion like the supernova in 1987 in the Magellenic Clouds. During that explosion, vast numbers of atoms were created from the hydrogen, helium, carbon and oxygen formerly in the star's interior. Another mechanism for creating heavy elements is two neutron stars colliding!
This happened some 5-6 billion years ago! ( Maybe longer.) All the evidence from material from Earth, the Moon, meteorites and data on the Sun points towards the Solar System forming 4.7 billion years ago. Since then most of the queer isotopes formed at that time have DISAPPEARED - DECAYED AWAY.
***This is because the ratio of neutrons to protons is too extreme in these isotopes and so the nuclei spontaneously change to create different atoms - they DECAY.
We say that the nuclei of such atoms are UNSTABLE and that the element is RADIOACTIVE.
The remaining nuclei after this period of time are either stable (unchanging) or take a long time to decay, for example Uranium-238, which has a half-life of 4.5 billion years.
There are exceptions. Cosmic rays continually bombard Earth, converting some Nitrogen-14 to the unstable Carbon-14. C-14 is taken up by all living organic material. ( A very recent discovery is that common lightning produces tiny amounts of C-14 on occasions!). Link to dating
In decaying , Uranium-238 gives rise to a chain ( a Decay Chain or Series ) of SHORT-LIVED
nuclei until a stable nucleus is formed. The short lived Radon-222 gas is formed
as part of the chain - a real danger to miners of uranium and to people who
live over some geological formations! The Radon gas is inhaled.
Radon decays to Polonium, giving off an α particle.
If the radon atom decays while in the lungs the α
radiation goes directly into the molecules of the lung. The decay to polonium
is followed by seven further steps in the
decay chain, of which three are α
emitters, two of which have half-lives measured in minutes or seconds (certainly
within the miner's normal life-time) and all further products are solids, thus
are likely to remain in the lungs. In the end, then radon decay can lead
to exposure to three α
particles, per atom decayed; not a healthy situation.
Link to Natural Types of Radioactivity