
1. Marie Curie is famous for her work in 1898, two years after Becquerels discovery, isolating radioactive elements unknown to humanity from "pitchblende". (Pitchblende is a mineral from the mountains of Germany on the Czech border.) This mineral contains uranium but the new elements were far more active than uranium and in smaller amounts. She died from radiation poisoning and even today her notebooks are dangerous.
She started with tonnes of pitchblende to obtain milligrams of new elements.
3. Balance the following equations:

4. The graph below represents the decay of Tc-99 with an initial activity of 50MBq and a half-life of 6.0 hours.
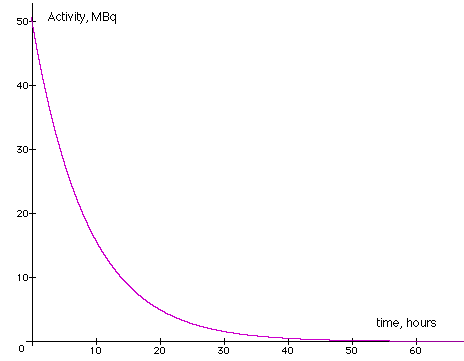
Plot the activity on the graph for 1, 2 and
3 half-lives.
The Tc-99 was created at 12.30 pm on Wednesday and is to be used at 6.30 am on Thursday morning, what activity can be expected when the sample is injected into the patient?
The patient is to be released into the public
when the activity has declined to 2MBq, when is the earliest that this
can take place?
5. Uranium-235 has a half-life of about 700 million years. If the age of the Solar System is 4700 million years, show that 6.7 half lives have passed.
From the above, by rounding off to 7 half lives, find the % fraction of the original U-235 remaining. (0.78%)
U-235 eventually decays into the isotope Pb-208 which is stable. A lead mine may still contain traces of the parent atoms from which it originated but very few of the other members of the Decay Series as they have short half lives. From the above calculation, what % fraction of metal is Pb-208 and what % fraction is U-235?
(fraction Pb = 99.22%, fraction U = 0.78%)