Go back to Background Radiation
Nuclear energy is the generated in two forms
Fusion means "joining together".
In a fusion process, Hydrogen nuclei are forced together to make Helium,
releasing energy.
The Sun uses H-1 , a temperature of 14 million 0C and a colossal density.Our best controlled attempts at fusion require a temperature of about 100 million 0C , the density of the gas exceeding that of water and held in controlled conditions for about 10 seconds. If we succeed in producing these conditions AND getting the energy out successfully , then we shall have a wonderful energy source . We haven't succeeded yet!
An "H-Bomb" uses H-3 (tritium) and the conditions generated by a uranium bomb!
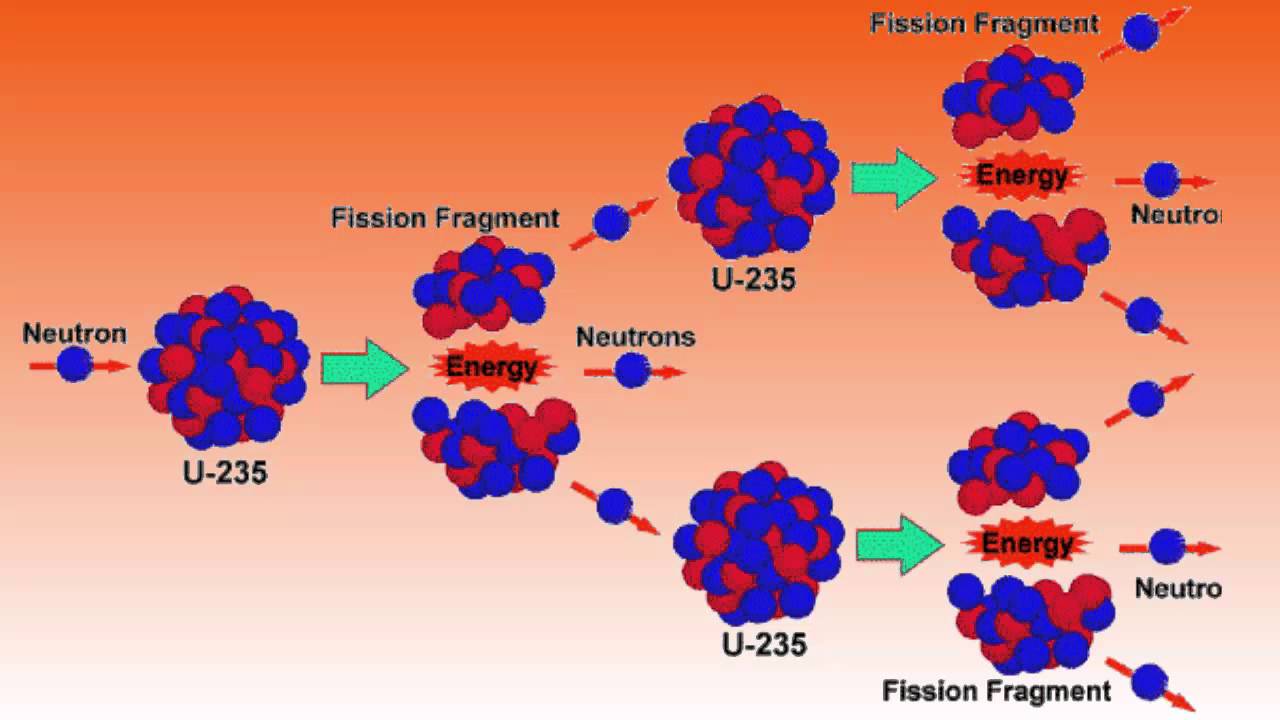
Fission means "splitting". Very large nuclei may be split down to smaller ones and give off energy. (This is because the most stable sized nuclei are those about the size of Iron!)
The best materials for this splitting process are U-235 and Pu-234. The uranium isotope is a rare isotope in uranium mines. (Uranium-238 is the major component.)
Using physical techniques it is possible to artificially raise the proportion of U-235 in a sample of the metal and make it suitable to sustain a "CHAIN REACTION".

The basic splitting reaction requires the use of slow moving neutrons. Fast moving neutrons don't have time to "stick" to a nucleus and split it . Indeed, the design of reactors incorporates a MODERATOR, a material present to slow down fast moving neutrons and thus increase the probability of causing the required reaction.
Three such reactions are;
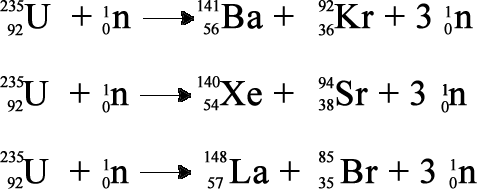
The produced neutrons continue the reaction by splitting another U-235. They are controlled so the reaction will not run away - go 'super-critical'. Criticality is achieved when the number of interacting neutrons is kept constant so the production rate is constant. This depends on the enrichment , the shape of the reactor, the absorbing control rods, the moderator and other factors.
The by-products are themselves usually radioactive
with varying half-lives. U-238, the most common isotope, tends to
decay to plutonium, and the surrounding material becomes radioactive.
(Note that the steel of the structure will slowly change to other elements
as time passes under the neutron bombardment compromising the integrity
of the structure with time! Engineers designing them must be careful!)