
MASS SPECTROMETERSnbsp; topics Magnetic particles
Machines for analysing the mass both of molecules and of the component atoms of the molecules. Mass spectrometers are the devices that showed decisively the existence of isotopes.
The basic technology is very old although both the control and the analysis devices attached now to the detectors are computerised.
Their history dates back to Sir JJ Thomson and his student Aston, in other words about 100 years. Many variations on mass spectrometers now exist with small, sensitive spectrometers making it onto chromatographs and space probes.
All depend on the ionization of the unknown material into gaseous ions, usually to positive, giving the ions a velocity and analysing them using magnetic or electrostatic fields.

University of Tasmania Central Laboratory's double focussing , magnetic sector Mass Spectrometer with a Quadrupole Mass Spectrometer at the exit.
Variations on the machines include the
Magnetic Sector including the Bainbridge Mass Spectrometer
The quadrapole mass spectrometers. These are often attached to capillary gas chromatographs.
Time of Flight spectrometers
Minor differences exist between machines used for organic and inorganic compounds. The range of an organic mass spectrometers is highly extensive. This machine is quite capable of measuring masses of 1000 amu. The University of Tasmania's Central Laboratory uses two magnetic sector machines and a variety of other mass spectrometers.
Magnetic Sector machines now tend to use a small accelerator sector with a PD of about 1500V immediately after the ions are introduced into the machine.
This ensures all ions have the same energy.
qV = 1/2 mv2 where q = charge = z e , V = accelerating potential difference, z = ionic charge, e = charge of electron
As they enter the magnetic field, they have a force acting on them of
qvB = mv2 / r thus v = qBr / m, substituting gives m / q = B2 r2 / 2 V
It follows that the mass is separated by radius but not linearly. Many machines then provide a variable magnetic field B to enable a wide range of masses to be identified.
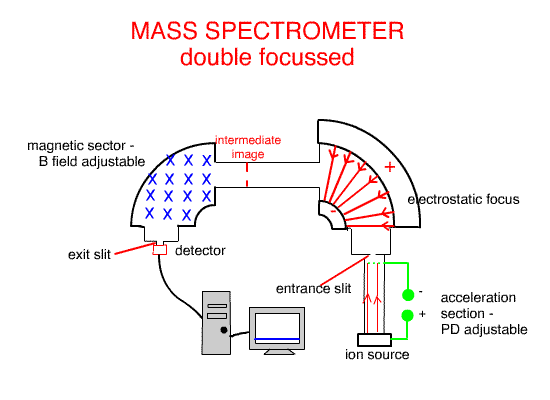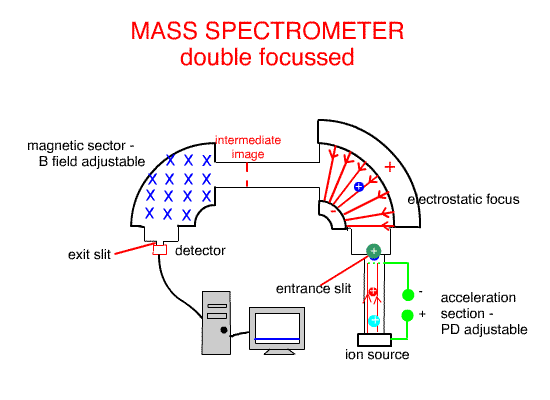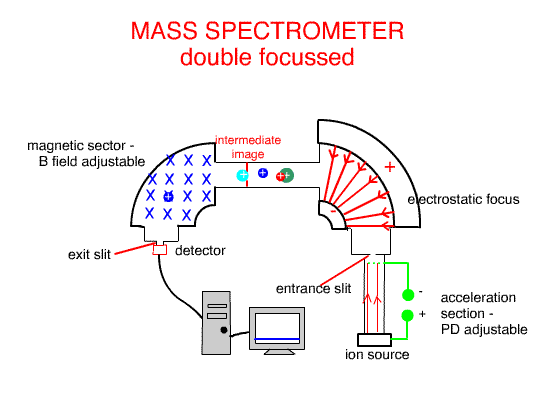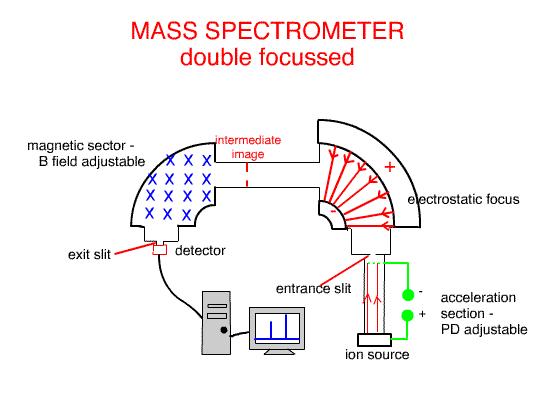
Using the Electrostatic focus to create a double focus machine, such as the one above, creates higher molecular resolution ( separation and identification ).
Sample size needed is only of the order of only nanograms ( 10-12 kg ).
Further analysis can be carried out using an attached quadruple machine at the exit slit, bypassing the prime detector.
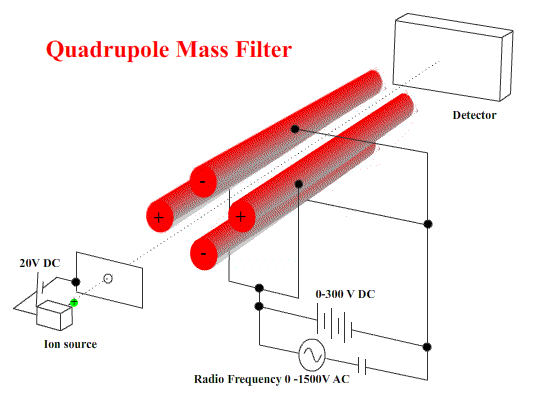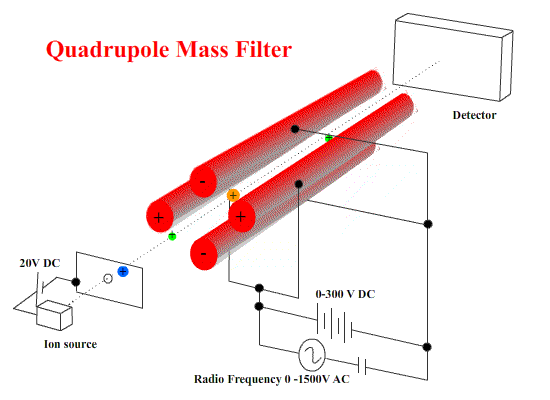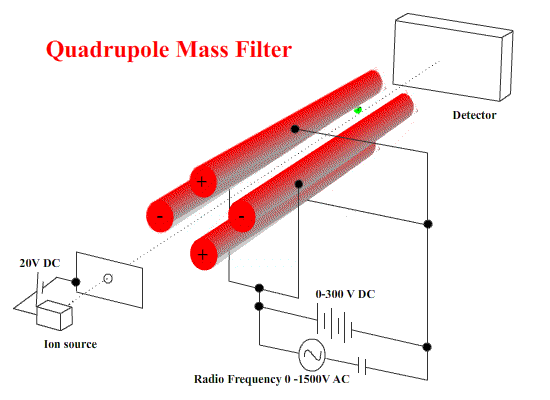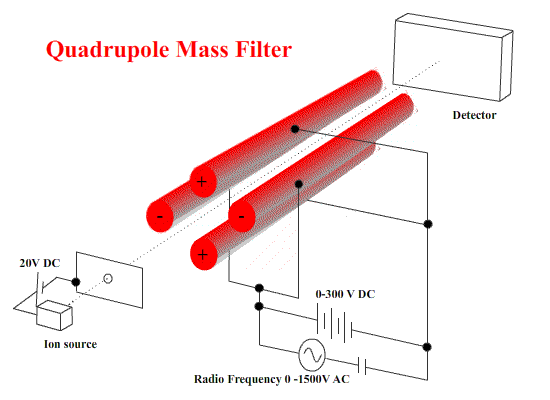
Quadrupole machines use electrostatics only. A combination of DC and AC fields on four stainless steel bars to cause ions to start wobbling - to move in irregular paths.
To select ions of a certain mass/charge ratio, the values of the DC and AC potential differences and the frequency are chosen so that the wobbles are small. They pass on to the detector. ( These ions are NOT resonating with the applied frequency.) Other ions WILL wobble - so badly that they strike one of the bars. ( These resonate with the applied frequency - small deviations build up to deflect the ion into the bar.)
These machines are small and fairly simple in construction.

Time of Flight machines accelerate pulses of ions to an equal energy then analyse them at a distant target simply by timing the ion.
qV = 1/2 mv2
thus v = ( 2qV/m )1/2 Simple "motion in a straight line" gives t = d ( m/2qV )1/2
The time depends on the square root of mass.
Pulses of ions are used to ensure that the ions start at equal times.
Source: Analytical Chemistry; Kellner et al Wiley-VCH