DE BROGLIE & HYDROGEN ATOM - FULL QUANTUM THEORY topics
1925 - de Broglie's electron waves applied to the Hydrogen atom make "clear" why only some orbits exist.
If we take Bohr's hypothesis and combine it with de Broglie's hypothesis then
mvr = n h / 2π ( Bohr )
but
mv = p = h /λ ( de Broglie )
substitute for mv
h /λ = n h / 2πr so n λ = 2πr n = 1, 2, 3 ......
WHOLE NUMBERS OF WAVES MUST FIT THE ORBIT ! WE HAVE STANDING WAVES AROUND THE CIRCUMFERENCE. MUSIC!
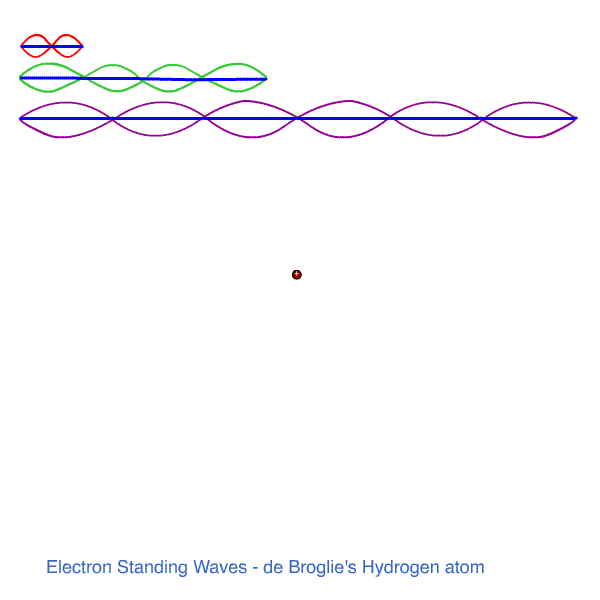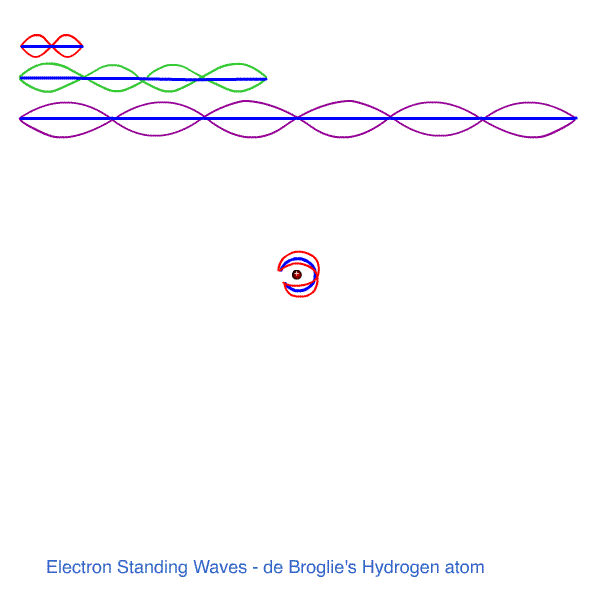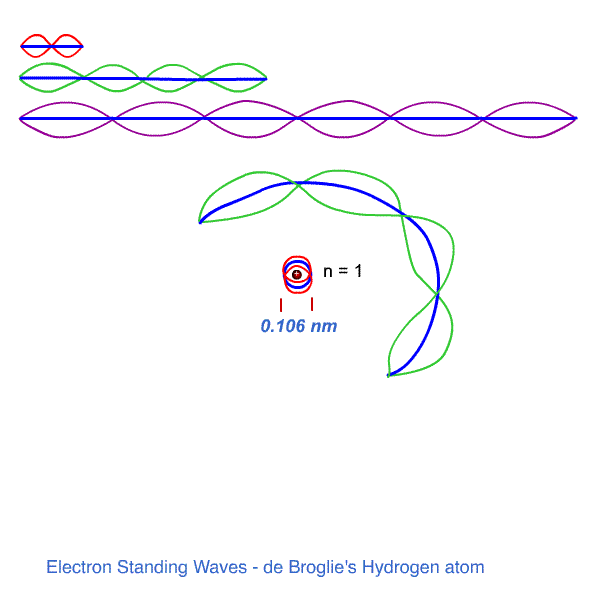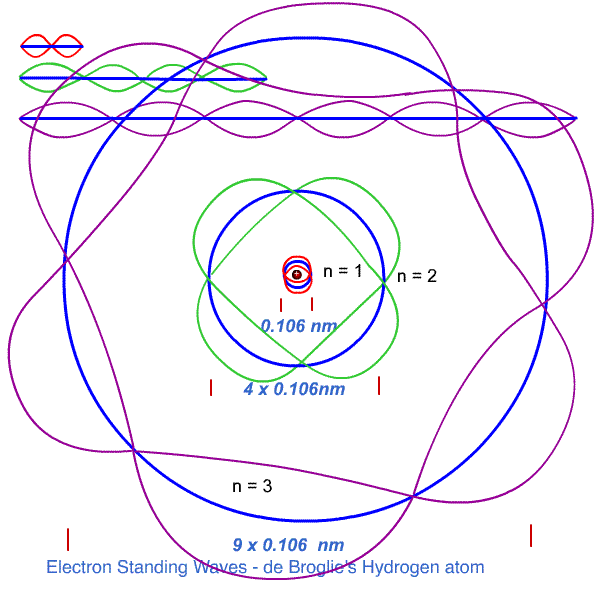
n = 1 , λ1 = 2πr1 - one wavelength fits the first orbit.
n = 2, 2λ2 = 2πr2 - two wavelengths fit the next orbit
n = 3, 3λ3 = 2πr3 - three wavelengths fit the third orbit
and so on.
The wavelengths of the various electrons are determined by their orbital momentum ( de Broglie ).

FOR ANY OTHER ORBIT, the momentum gives a wavelength which does not match the corresponding circumference exactly.
The waves, in not matching precisely the circumference, end up, by superpositioning, exactly cancelling itself as everywhere on the circumference eventually the oppositely shaped wave will cancel the wave.
QUANTUM MECHANICS - AN INCREDIBLY SUCCESSFUL THEORY ( no one understands ).
In 2001, 37% of the US economy was based on products derived from the understanding of quantum mechanics. This includes computers and biochemistry.
Two areas are largely developed. Quantum Electrodynamics - the full quantum theory of charge and light. This is VERY successful and covers atoms, reflection, crystal structures, molecules, superconductivity and all of chemistry and hence biochemistry.
Quantum Chromodynamics is the theory of the nucleus and subatomic particles. This is less well advanced than the above and is the experimental area that the big accelerators operate in. Increasingly, this theory is being integrated with cosmology both in observation and in advanced theories trying to integrate gravity and quantum physics.
First versions were developed in
Schrodinger's version is the most quoted.
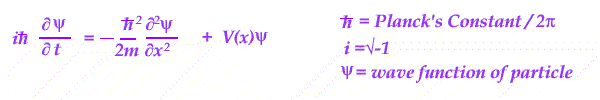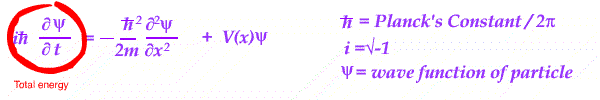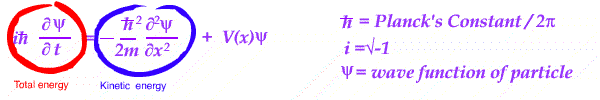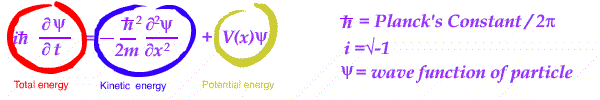
Essentially an equation is set up in energy - kinetic and potential, for a wave in position and time.
With different energy conditions, the equation is solved and the resulting wave is interpreted.
Its wave shape is treated as a measure of the PROBABILITY of finding a given charged particle in the system analyzed.
When applied to atoms, the standing waves of electrons that result in 3-D are in weird directions and shapes. This accounts for the shapes and energy levels of chemical bonds hence molecular shapes and structures. Software that carries out these calculations is now available even for desktop computers!
Heisenberg's approached is based on matrix mathematics and out of fact that A.B does not always = B.A for matrices comes one of the most famous statements of quantum mechanics -
Heisenberg's Uncertainty Principle
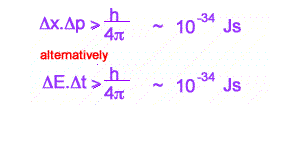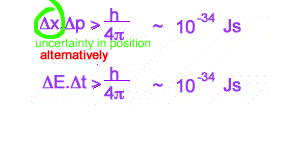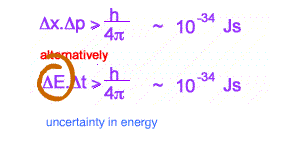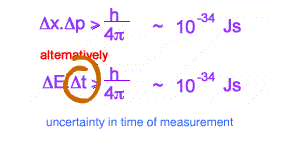
This inequality is used as a reason for some of the bizarre behaviour of the tiny world, eg Single and Double Slit behaviour for electrons. Increasingly it is becoming clear that more profound oddities exist in interpreting quantum mechanics. Many versions of the inequality exist. The above are the best known.
The first version quoted can be used to look at difficulties in measuring the very small. Simply, when attempting to measure very small distances, with small uncertainties in position, very large changes in momentum of the particle of unknown size or direction can be expected. At the very small, you cannot known simultaneously position and momentum!
The second version relates to measured energy and the time spent to measure this energy. IF the time is small enough, with tiny uncertainty, large fluctuations in energy can be expected . These fluctuations in energy can exist as mass ( by E = mc2 )- what are called VIRTUAL PARTICLES.
Virtual particles are used to explain forces at a distance that Newton could not explain but used.
Electromagnetic forces ( electrostatics and magnetism ) are explained by exchanging photons which have momentum, at the "virtual" level - ie cannot be seen. This is Quantum Electrodynamics.
Gravity is explained by a hypothetical particle called the "graviton".
The strong nuclear force by the "gluon" . There are 11 of these and they have all been created in accelerators.
and the weak nuclear force by the "weakon" family. Again, all have been created in accelerators from 1984. 3 exist, W+ , W- and Z0
Virtual particles' effects have been measured! Atoms and the vacuum of space are swarming with virtual particles!
Excellent reading includes
"In Search of Schrodinger's Cat" by John Gribbin
"Schrodinger's Kittens" also by John Gribbin
"Q.E.D" by Richard Feynman, Princeton Press