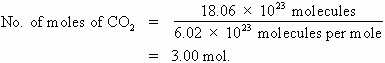
Relative Molecular Mass
A similar relative scale to the atomic mass scale could be developed for gases using masses of equal volumes of gases as a base for comparison. As gases exist as individual molecules, such a scale would be termed the relative molecular mass scale.
As before, a parallel scale measuring mass in grams will link to moles of gases.
For example:1 mole of hydrogen gas contains 6.02 x 1023 H2 molecules
1 mole of carbon dioxide gas contains 6.02 x 1023 CO2 molecules.
We shall see shortly that the relative molecular mass and relative atomic mass scale can in fact be thought of as a single scale. We will also be able to include masses of ions on such a scale.
Example 1:
How many moles of carbon dioxide gas will you have if you are told that you have 18.06 x 1023 molecules of the gas in your breath?
Solution:
First we must remember that there are 6.02 x 1023 particles in 1 mole of any substance. So we can write this problem as a division:
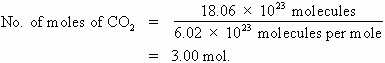
We can also relate the number of moles of a substance to the mass of the substance using the relationship between the mole of a substance and the relative atomic (or molecular) mass.
Example 2:
How many moles of hydrogen gas will be produced from a reaction that evolved 40 grams of the gas? (Molecular mass of hydrogen gas, H2 = 2.0.)
Solution:
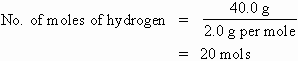
Example 3:
An oxygen cylinder contains 6.0 moles of gas. If one mole of oxygen gas has a mass 32.0g, what will be the mass of the gas in the cylinder?
Solution:
Mass of oxygen = 6.0 mol. x 32.0 g per mole
= 192.0 g
Defining and relating the terms
To formalise and extend our understanding of relative atomic mass and the mole we can give a definition based on our modern understanding of the atomic mass scale.
The relative atomic mass of an element is the ratio of the average mass of the atoms of the elements compared to the carbon−12 isotope that is allocated the mass of 12 units.
A mole of a substance is equivalent in mass to the relative atomic mass or relative molecular mass in grams.
The molar mass is the mass of one mole of a substance.
Now try these problems.