Mass-mass problem
further examples
Close window when finished.
All mass-mass problems can be solved by following these steps:
Example 4:
Under severe conditions; high temperature, pressure and concentration; hydrogen gas and nitrogen gas can be coerced into forming ammonia, NH3, also a gas at these temperatures.
What mass of N2 would be required if mixed with excess hydrogen gas (meaning more than enough - the reaction will cease when the nitrogen runs out) to produce 850g ammonia? What mass of H2 would be used?
Solution
One mole of ammonia contains:
1 mole of N
3 mole of H.
|
1 mole of nitrogen is |
14g |
||
|
1 mole of hydrogen is |
1g, so |
3 mole is |
3g |
|
1 mole of NH3 is |
17g |
Nitrogen and hydrogen are both diatomic. One mole of nitrogen gas is 28 grams, one mole of hydrogen gas is 2 grams.
Step 1. Write a balanced equation. N2(g) + 3H2(g) → 2NH3(g)
Step 2. ![]() , so
850g of ammonia is 50 mole
, so
850g of ammonia is 50 mole
Step 3.
![]() ,
50 mol of ammonia require 25 mole
of nitrogen
,
50 mol of ammonia require 25 mole
of nitrogen
Step 4.
![]() ,
so 25 mole of nitrogen gas is 700g.
,
so 25 mole of nitrogen gas is 700g.
Repeat for hydrogen:
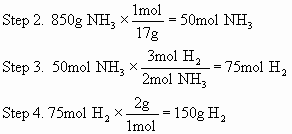
700g + 150g = 850g; this would suggest our calculations make sense.
Example 5: This one is a fair bit curlier, because although the steps come in along the way, the nature of the problem requires they be un-staightforward.
When poured together, what mass of precipitate will result? (Assume the resulting precipitate is completely insoluble in water.)
Solution
Step 1:
|
The reaction equation: |
NaCl + AgNO3 → AgCl + NaNO3 |
|
The ionic equation: |
Na+(aq)+Cl-(aq)+Ag+(aq)+NO3-(aq)→ AgCl(s)+Na+(aq)+NO3-(aq) |
|
The spectators: |
Na+(aq) and NO3-(aq) |
|
The net ionic equation: |
Ag+(aq) + Cl-(aq) → AgCl(s) |
Step 2:
![]() , which
dissolved yields 1.711mol Na+ and 1.711mol Cl-.
, which
dissolved yields 1.711mol Na+ and 1.711mol Cl-.
One mole of AgNO3 contains:
1 mole of Ag
1 mole of N
3 mole of O.
|
1 mole of silver is |
107.870 g |
||
|
1 mole of nitrogen is |
14.0067g |
||
|
1 mole of oxygen is |
15.9994g, so |
3 mole is |
47.9982g |
|
1 mole of AgNO3 is |
169.875 g |
![]()
which dissolved yields 0.589mol Ag+ and 0.589molNO3-.
Step 3:
We haven’t got one mole of Ag+. We have only 0.589 mole. When 0.589 mole of Ag+ have reacted it will be all used up. The reaction will stop! Thus:
0.589mol Ag+(aq) + 0.589mol Cl-(aq) → 0.589mol AgCl(s)
Step 4:
One mole of AgCl contains:
1 mole of Ag
1 mole of Cl.
|
1 mole of silver is |
107.870g |
|
1 mole of chloride is |
35.453g |
|
1 mole of AgCl is |
143.323g |
![]() 84.42g precipitate out of solution.
84.42g precipitate out of solution.
Further discussion:
If we now pour off the remaining solution with the sodium, nitrate and excess chloride ions we can evaporate the water and weigh the dry contents.
![]()
![]()
Of the initial 1.711mol Cl- 0.589mol have been removed by silver. That leaves 1.122mol.
![]()
84.42 + 39.34 + 36.52 + 39.78 = 200.06g, the same total mass we started with, after you allow for rounding errors. Mass is conserved in chemical reactions.
If we could separate the crystals we would find 1.122mol NaCl(s) and 0.589mol NaNO3(s).
Notice in this problem the following algorithm - a series of four steps which produces the correct solution. (No, computers aren’t the only beasts who can execute algorithms.)
REMEMBER: Substances react on a particle-to-particle basis. This means, in the case of ammonia, one nitrogen molecule reacts with three hydrogen molecules to produce two ammonia molecules, or one mole of nitrogen reacts with three moles of hydrogen to produce two moles of ammonia. This negates the idea that, say, one gram of nitrogen reacts with three grams of hydrogen to produce two grams of ammonia, or one litre of nitrogen reacts with three litres of hydrogen to produce two litres of ammonia. It’s number, not mass or volume.
In the same way you always have to find time for problems of motion in two directions, so you must always find the number of moles to solve mass-mass problems.
Now try some mass-mass problems.
Or close window to return to main thread.