Isotopes
(and weighted averages)
Previous Page: The problem with atoms
In our work earlier this year, we suggested that the mass of an individual atom was simply the number of protons plus the number of neutrons in the nucleus (since the electrons have negligible relative mass). This quantity was called the mass number, and was acknowledged as being a whole number. But when we examine the relative atomic mass of an element such as chlorine, in the periodic table, we immediately identify a problem. The value listed is 35.5, which is nowhere near a whole number. But we know that not all atoms of an element are identical. Many naturally occurring elements have more than one type of atom present in any sample and these atoms have different mass numbers.
Different kinds of atoms of the same element are called isotopes. (You will have come across isotopes if you studied the unit on radioactivity.) Isotopes have the same number of protons and electrons but a different number of neutrons. The atomic mass of an element could then be said to be the average mass of all isotopes present in the sample. This partially explains the fractional values in the periodic table.
Example
Calculate the relative atomic mass of chlorine using the following information: Chlorine has two isotopes, chlorine−35 and chlorine−37. The percentage of chlorine−35 is 75%, that of chlorine−37 is 25%.
![]()
Therefore,
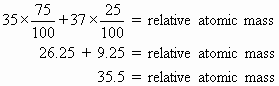
Compare this result with the figure given in the periodic table.
If I weigh 5 coins each with a mass of 6 grams and 5 coins each with a mass of 7 grams, the average mass of the coins is 6.5 grams. If I weigh 6 coins each with a mass of 6 grams and 4 coins each with a mass of 7 grams, the average mass of the coins is not 6.5 grams.
Because I have repeating values, I can use a weighted average to find the average in the second case. To do this I find the average of 6+6+6+6+6+6+7+7+7+7. I can add all these up, or I can say "that's 6 × 6 + 4 × 7" which is 36 + 28 which equals 64 grams. Since there are ten coins, the average is the total divided by the count, or 6.4g.
Example: Looking at this problem from the other direction, say I am given 946 coins that are a mixture of the two type of coins listed above and asked to find the percentage of each in the sample.
Solution: I might decide to take a short cut - I weigh them - and find the total mass to be 6097 grams. That means the average mass is 6097/946 = 6.445g. I then decide that I'm going to let c represent the percent of 6g coins and 100-c be the percent of 7g coins. Thus
| 6.445 | = (6 × c/100) + (7 × [100-c]/100) |
| = (0.06c) + (7 - 0.07c) | |
| = 7 - 0.01c | |
| 0.01c | = 0.555 |
| c | = 55.5% |
Since c stood for the percent of 6g coins 55.5% of 946 coins, or 525 coins weigh 6 grams.
Now try these problems.