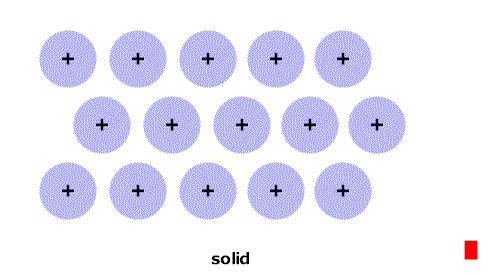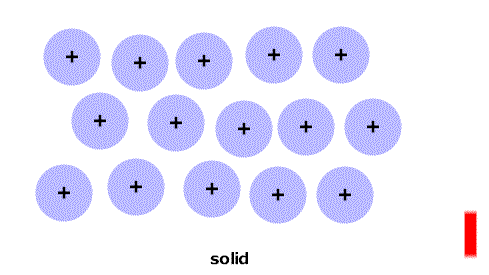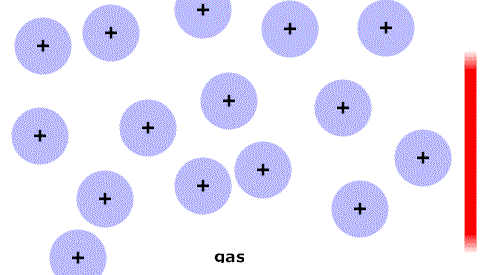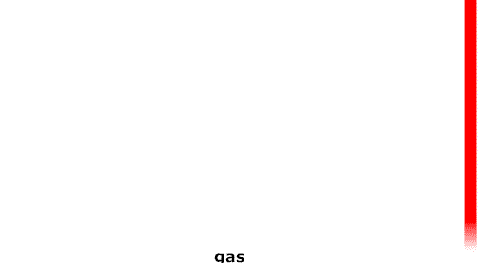
As the temperature increases the particles have more energy and are more active.
At particular temperatures some of the attractions between them are overcome and the state changes: solid->liquid->gas.
(Think of the red stripe as a thermometer.)
High melting temperatures.
Close window when finished.Similar to hardness, the melting point of a substance reflects how strongly the particles are held in place in their lattice. More precisely, it indicates how much energy is required to break the lattice down. If more energy is required the melting point will be higher.
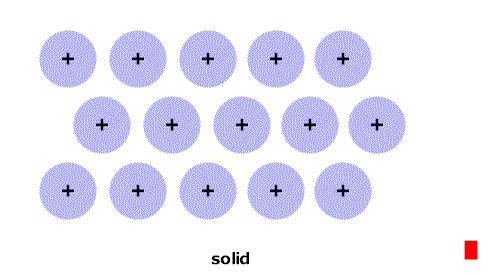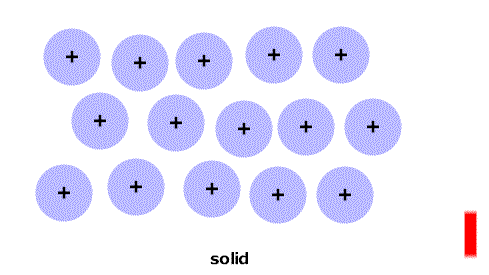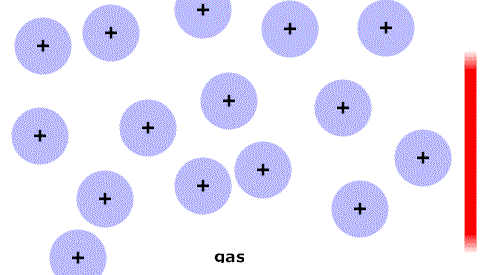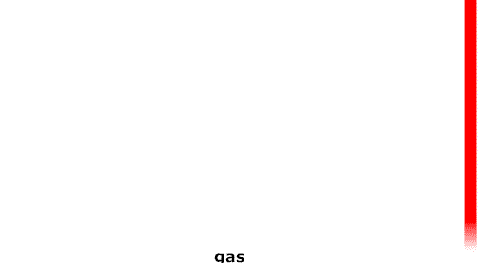 |
As the temperature increases the particles have more energy and are more active. At particular temperatures some of the attractions between them are overcome and the state changes: solid->liquid->gas. (Think of the red stripe as a thermometer.) |
As we just saw, metal ions held in place by the electrostatic attraction of the delocalised electrons can shift around a bit without undue effort. Thus metals have relatively low melting temperatures, that is, low compared to ionic compounds and covalent networks.
Close window to continue.