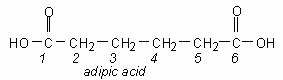
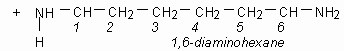
PreviousPage: Addition Polymerisation
The other main type of polymer formation is called condensation. The name indicates that in the process of forming the polymer, another product also occurs, usually water. As addition reactions are also called chain-reactions, so condensation reactions are also called step-reactions. Go figure.
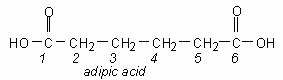
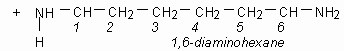
| → | 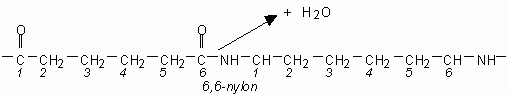 |
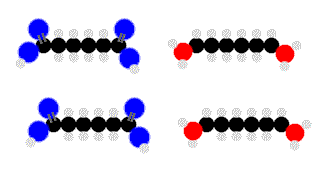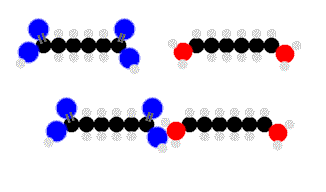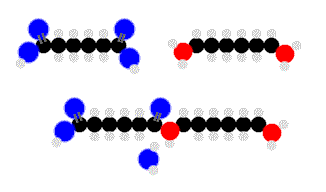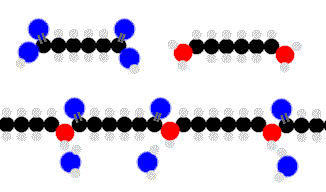
Above we see adipic acid (left)and 1,6-diaminohexane condensing to form 6,6-nylon. One water moleculeresults from each linkage.
In this reaction every linkage produces a water molecule. Every condensation linkage produces some sort of simple product, though it need not be water.
Proteins are condensation-linked amino acids.
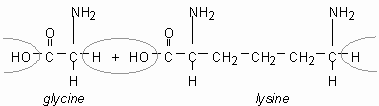
| → | 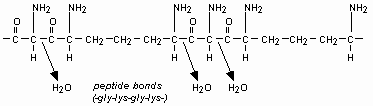 |
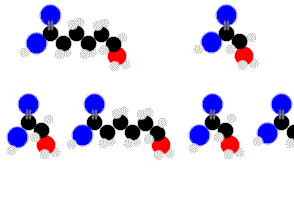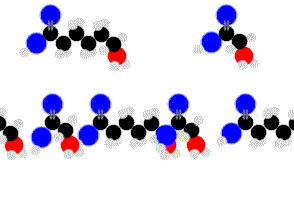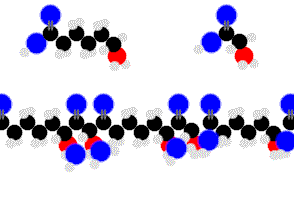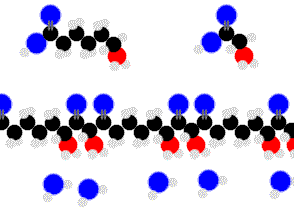
Finally we are getting something complex enough to sustain life.
Unlike in addition reactions, in condensation reactions the different monomers do need to alternate regularly, unless they have identical functional groups.
Common addition polymers are: