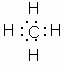
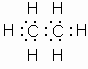The simplest carbon compound is methane, CH4.
|
|
H | H-C-H | H |
 |
Electron dot diagram, expanded structural formula and atom model for methane.
Two methanes may be joined to form ethane, C2H6. Three make propane, C3H8. Four give butane, C4H10.
|
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Electron dot diagram
and condensed and expanded structural formula for ethane. Butane
is far right.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 |
 |
 |
 |
| Two alternate models of ethane (left) and butane. | |
Note two things: first, all these molecules have the general molecular formula CnH(2n+2). Second, all these names end in -ane. The suffix -ane is used to designate chains of carbon with all single bonds. In the same way that fluorine, chlorine, bromine and iodine are grouped together and called, collectively, halogens, so carbon chains with single bonds are called alkanes.
Problem 1. What would be the formulae for alkane chains 5, 6, 8 and 20 carbons long?
Alkanes are a sub-group of the larger group of hydrocarbons. Hydrocarbons are molecules containing only hydrogen and carbon.
Table 1, below, shows the prefixes for the first ten hydrocarbon chains. You may have heard of formic acid. It causes the itch when an ant bites. The name formic tells you that it is an organic acid with a carbon chain one carbon long:
|
HC-OH || O |
 |
|
Structural formula for formic (methanoic) acid. |
|
|
Branch |
Systematic name |
Traditional name |
|
-CH3 |
Meth- |
Form- |
|
-C2H5 |
Eth- |
Acet- |
|
-C3H7 |
Prop- |
|
|
-C4H9 |
But- |
|
|
-C5H11 |
Pent- |
Am- |
|
-C6H13 |
Hex- |
|
|
-C7H15 |
Hept- |
|
|
-C8H17 |
Oct- |
|
|
-C9H19 |
Non- |
|
|
-C10H21 |
Dec- |
Table 1. Names of shorter hydrocarbon chains.
Likewise, acetic acid, the active ingredient (and identifiable smell) in vinegar, it is an organic acid with a carbon chain two carbon long:
|
CH3-C-OH || O |
 |
 |
| Structural formula and models of acetic (ethanoic) acid. |
||
In both cases we are dealing with traditional names. In current literature you would expect to find the names methanoic acid and ethanoic acid used instead, based on the meth- and eth- prefixes in the table.
 |
Woof! |
Problem 2: Draw the structural formula for butanoic acid.
Next page: Alkenes & Alkynes